Deep within every one of your cells, a remarkable molecular machine operates with breathtaking speed and precision. Hundreds to thousands of crucial molecules shuttle every second through thousands of gateways called nuclear pores. These pores are the sole entry and exit points for the cell's nucleus, the command center containing our genetic blueprint. For decades, scientists have puzzled over how this transport system achieves such exquisite selectivity while maintaining incredible throughput. New high-definition research now reveals the surprising answer: controlled chaos.
The Gateway to the Genetic Library
The nucleus is the most exclusive organelle in the cell, housing the DNA that directs all cellular activities. To protect this vital information, it is surrounded by a double membrane. The only passages through this barrier are the nuclear pore complexes—massive assemblies of proteins that act as both gate and gatekeeper. They must allow essential molecules like transcription factors and signaling proteins to enter, while permitting RNA and ribosomal subunits to exit, all while blocking unauthorized traffic. The scale of this operation is staggering, with each pore processing a continuous stream of molecular cargo.
A Jumble of Proteins with a Purpose
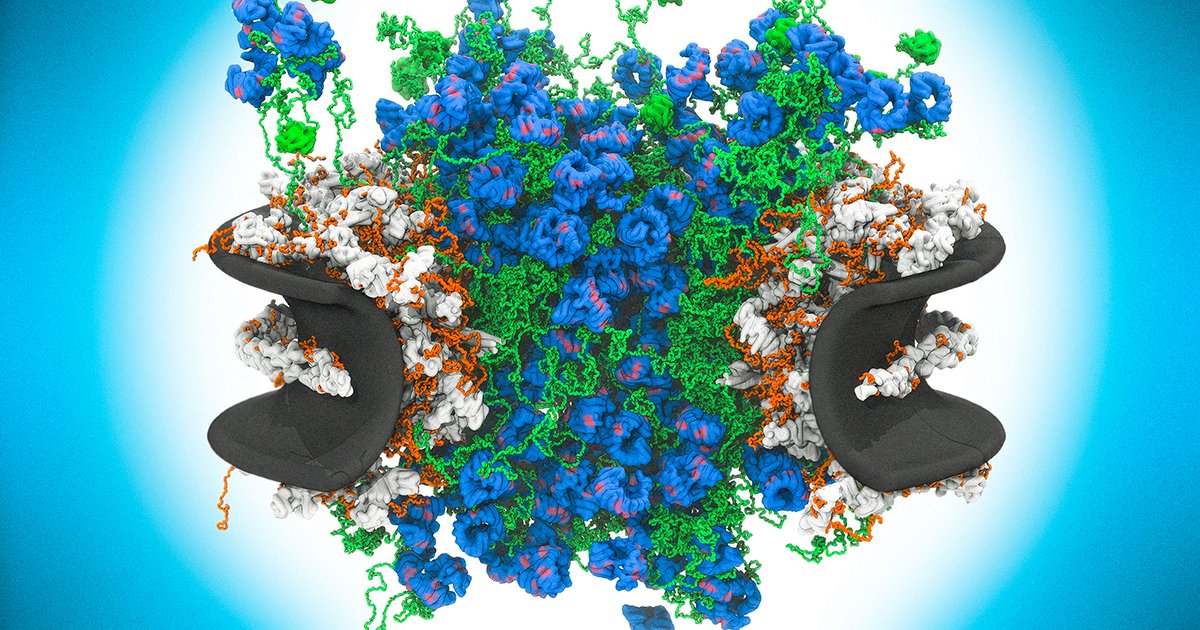
For years, the structure of the nuclear pore's central channel appeared confusing. Instead of a neat, orderly arrangement, it was filled with a seemingly tangled mass of proteins called FG-nucleoporins. These proteins contain long, flexible regions rich in the amino acids phenylalanine and glycine (the "FG" in their name). Early models struggled to explain how this apparent molecular jumble could function as a selective filter. The new research, employing cutting-edge imaging techniques, has finally captured this dynamic structure in action, transforming our understanding from static architecture to functional disorder.
The study shows that these FG-nucleoporins are not static bouncers but dynamic, ever-moving sentinels. They create a dense, gel-like mesh within the pore channel. Authorized cargo molecules carry a specific molecular key—a nuclear localization signal—that allows them to interact transiently with the FG-repeats. This interaction is like a handshake that lets the cargo momentarily part the tangled mesh and slip through. Unauthorized molecules, lacking the correct key, find the mesh an impenetrable barrier. The system's efficiency stems from its disorder; the constant motion and flexibility allow rapid binding and release, enabling high-speed transport without compromising security.
Disorder as a Design Principle
This discovery challenges the traditional view that biological machines require rigid, ordered structures to function precisely. Instead, the nuclear pore complex leverages intrinsic disorder as a core operational feature. The floppy, unstructured regions of the FG-nucleoporins are not a bug but the central feature of the design. Their constant thermal motion prevents them from forming a permanent, solid plug, while their collective density creates a selective barrier. This principle of functional disorder may be a recurring theme in biology, where flexibility and adaptability are paramount. It echoes findings in other fields, such as the way natural fracture patterns in tissues use controlled chaos for strength and growth.
The implications extend beyond cell biology. Understanding how natural systems achieve robust function from disordered components can inspire new approaches in nanotechnology and materials science. For instance, engineers designing synthetic filters or molecular sorting devices might look to this biological model of a gate that is both open and closed simultaneously. Furthermore, the sophisticated logic of this cellular gateway offers a humbling contrast to current technological challenges, such as why humanoid robots struggle with basic physical tasks that biology performs effortlessly.
A New Window into Cellular Function and Disease
Visualizing the pore complex in such detail provides more than just a beautiful image; it offers a functional blueprint. Mutations in nucleoporin genes are linked to a variety of human diseases, including certain forms of heart disease, neurological disorders, and cancers. By understanding precisely how the normal pore operates, researchers can begin to decipher how these mutations disrupt molecular traffic, leading to cellular dysfunction. This high-definition view acts as a baseline for diagnosing what goes wrong when the cellular gatekeepers fail.
The research also highlights the accelerating synergy between biology and computational analysis. The data required to model such a dynamic, disordered system is immense, relying on advanced imaging and complex simulations. This interplay mirrors progress in other disciplines, where AI is uncovering hidden patterns and computational verification is reshaping discovery. As tools improve, we can expect even clearer views of life's most intricate machinery, revealing that what once looked like chaos is, in fact, a highly evolved and elegant form of order.
Ultimately, the nuclear pore complex stands as a testament to evolution's ingenuity. It solves a critical logistical problem—secure, high-volume transport—not with rigid clockwork but with a fluid, intelligent filter. This revelation reminds us that in the molecular world, perfection often lies not in stillness and symmetry, but in purposeful and dynamic disorder.